| By: Gary Greenberg | Posted: 7/1/2001 |
Page Views: 3009 | |
| You have to accept that there's a structural change in your brain when you take drugs like Prozac. | |||

I think you have to accept that there's a structural change in
your brain when you take drugs like Prozac.
Psychotherapists love to argue. We argue about treatment theories, about our clients and their families, about the office coffeepot. And during the past decade we have tended to fixate, as we say in the business, on the subject of Prozac. It used to be fairly easy to agree about commonly prescribed psychiatric drugs such as Valium: They anesthetized people, covered up problems, illegitimately took the place of therapy. But Prozac and the other antidepressants that work by enhancing serotonin activity in the brain have eluded such easy criticism. Often we would find that our clients who took them felt more alive, more resilient, more able to engage in the honest self-reflection necessary to therapy. And we could not help but agree with Peter Kramer, who wrote in Listening to Prozac that the drug can remake the self— which was supposed to be our job.


Therapists haven't been alone in their Prozac anxiety. Americans have always
been ambivalent about mind-altering drugs, and many wonder if it is a good thing
that today some 30 million Americans—many of them not clinically depressed but
rather among the worried well
—have taken serotonin enhancers at one
time or another. But other issues are more troubling, like the serious side
effects—which include violent impulses, agitation, and sexual dysfunction—that
have been reported since the drugs first appeared and have never been fully
confirmed or disproved. What's perhaps most disconcerting is the fact that 15
years after the first of the serotonin enhancers—Prozac—was put on the market,
the precise reason why they relieve depression remains unknown.

Some scientists, however, think they are on the verge of solving this mystery, suggesting that serotonin enhancers may work by encouraging the growth of new brain cells. At the same time, other researchers have found that high doses of these drugs cause changes in neurons that some would call brain damage—a finding that may have some bearing on the range of reported side effects. And both sets of research point to the possibility that serotonin enhancers alter brains in ways researchers never imagined.
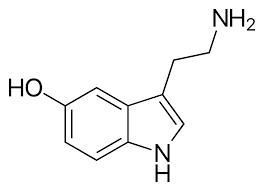
Serotonin, also known as 5-hydroxytryptamine (5-HT), was first isolated in
1933, when it was discovered in the gut and called enteramine. In 1947 it was
found in blood platelets, and the molecule earned its current name, serotonin,
when it also proved to constrict blood vessels. Soon after, serotonin was
 identified in the brain. But its role was unknown until some drug tests in the
1950s drew unexpected results. Researchers found that three drugs—isoniazid and
iproniazid, both antituberculars, and imipramine, an antihistamine—improved the
moods of test subjects. At around this time, clinical discoveries and
technological advances were turning a once heretical idea—that nerve impulses
in the brain were transmitted chemically—into orthodoxy. So the unanticipated
psychoactive effects in the drug tests prompted scientists to study how these
compounds affected chemical messengers in the brain called neurotransmitters.
They found that all three agents acted on a group of neurotransmitters known as
the monoamines. From this, they concluded that monoamines must be important in
depression. The clinical implications of this discovery were not lost on
scientists at pharmaceutical companies, and in 1975 a group at Eli Lilly quietly
reported that they had synthesized 110140, a substance that targeted serotonin
with precision. Eleven years later, 110140 became Prozac, one of the most
successful drugs ever brought to market, responsible in 1999 for 26 percent of
the revenues of one of the largest companies in the United States.
identified in the brain. But its role was unknown until some drug tests in the
1950s drew unexpected results. Researchers found that three drugs—isoniazid and
iproniazid, both antituberculars, and imipramine, an antihistamine—improved the
moods of test subjects. At around this time, clinical discoveries and
technological advances were turning a once heretical idea—that nerve impulses
in the brain were transmitted chemically—into orthodoxy. So the unanticipated
psychoactive effects in the drug tests prompted scientists to study how these
compounds affected chemical messengers in the brain called neurotransmitters.
They found that all three agents acted on a group of neurotransmitters known as
the monoamines. From this, they concluded that monoamines must be important in
depression. The clinical implications of this discovery were not lost on
scientists at pharmaceutical companies, and in 1975 a group at Eli Lilly quietly
reported that they had synthesized 110140, a substance that targeted serotonin
with precision. Eleven years later, 110140 became Prozac, one of the most
successful drugs ever brought to market, responsible in 1999 for 26 percent of
the revenues of one of the largest companies in the United States.
Yet a crucial question remains: We simply don't know why having a synaptic
lake brimming with serotonin makes people happier. While there is evidence that
some depressed people have lower levels of serotonin breakdown products in their
spinal fluid and different brain anatomies from the overall population, the
proof of the commonly held notion that a deficiency or imbalance in the
serotonin system causes depression remains weak. Nor is it known why the drugs
generally take three to six weeks to alter mood, why they help people with
nondepression-related problems like shyness or compulsiveness, why people who
were not depressed in the first place sometimes feel better than
well,
or why the drugs sometimes lose their efficacy over the long term.
Despite gaps in our knowledge, the post-Prozac era has seen the rise of a singular idea, one that can be called mythic for both its explanatory power and its lack of evidence—depression is best understood and treated as a biochemical aberration for which drugs like Prozac are the silver bullets.

You hear that pop? I never get tired of listening to that!
Barry
Jacobs, professor of psychology at Princeton University, steers me toward the
source of the sound, a rack of electronic equipment in his lab that includes an
amplifier and speaker and an oscilloscope, whose green line leaps into action
with every pop. In a room behind us, a yellow cat rests his head on the edge of
his cushioned box. He looks like any other contented house cat but for the
thicket of tiny wires that runs from his head to a boom on the ceiling and then
out to the oscilloscope and other measuring devices. Despite its
cobbled-together look, this apparatus is performing a precise and delicate task:
The wires terminate in hair-thin probes, one of which measures activity in a
serotonin-releasing neuron in the cat's brain stem. Every time the neuron fires
and sends serotonin toward another cell, the action registers on Jacobs's
machines. Because the cat is free to move about during the experiment, Jacobs
can correlate serotonergic activity with behavior.
Jacobs's years of work with cats like this one have earned him the nickname Mr. Serotonin. So it was only natural that the emergence of serotonin-enhancing drugs would get him interested in depression. But it was a casual conversation with his office neighbor, neuroscientist Elizabeth Gould, that put him onto his new theory of how the drugs work. Over the past few years, Gould's research has shown that adult monkeys routinely grow new brain cells, a process known as neurogenesis. Her work has given support to an emerging body of evidence that people, too, undergo neurogenesis throughout their lives. The discovery is provocative because neurogenesis seems most prevalent in the hippocampus—a region of the brain associated with learning, memory, and, perhaps, emotion.
We began talking,
Jacobs recalled, and [Elizabeth] says:
'What do you think serotonin's impact would be on neurogenesis?' I said, 'I
don't know. It's a good question. Why don't we try?' So we did these very simple
experiments.
Jacobs and his lab team injected rats with a drug that
attaches to DNA in cells that are about to divide. The compound effectively
labels cells born after the drug was given. The results were impressive: Rats
given Prozac made 70 percent more neurons than the rats that hadn't received the
drug.
Jacobs's bright blue eyes stand out even more than usual against his white
hair and beard when he remembers what happened next. As soon as we showed
that serotonin could promote neurogenesis, immediately I said, 'This could have
implications for depression.' In fact, this little finding provides as good a
theory of depression as anything else that's out there.
That theory,
simply stated, is that depression is linked to neurogenesis. In a depressed
patient, the brain stops making new neurons; when neurogenesis resumes, the
depression lifts.

Psychologists have found that stress can often trigger depression. And stress
floods the brain with certain hormones (glucocorticoids) that are known to
suppress neurogenesis or even kill neurons, especially in an area of the
hippocampus known as the dentate gyrus. Studies have found that depressed
patients have somewhat smaller hippocampi than nondepressed people. Moreover,
patients with diseases like Cushing's syndrome and temporal lobe epilepsy that
cause cell loss in the hippocampus have a much higher risk of depression than
the rest of the population. And it takes about three to six weeks for new cells
to mature—the same time it takes serotonin-enhancing drugs to make a difference
in a patient. Add all this evidence up and you have, in Jacobs's view, the
leading candidate
for understanding what happens in the brains of
depressed people and why drugs like Prozac help them.
Scientists at Yale's Laboratory of Molecular Psychiatry, led by Jessica
Malberg, have tested this hypothesis and shown that a wide range of
antidepressant treatments—electroshock therapy, serotonin-enhancing drugs, and
other types of medications—will increase neurogenesis in rats. Malberg cautions
that we don't understand the relationship between neurogenesis and the effects
of antidepressants. There's absolutely a correlation,
she says,
but we don't know that it's definitely neurogenesis that's [relieving
depression].
Jacobs also sounds a note of caution. He warns that we know so little
about mood and the neural basis of it,
but he is willing to speculate on
how neurogenesis may help lift depression. Perhaps, he says, people get
depressed when chronic or acute stress brings about the death of neurons
or the failure to grow new neurons. People dwell on negative things and are
incapable of forming new cognition about the future being positive and things
getting better—until they have the ability to grow new neurons that mediate
this new cognition.
While nobody knows for sure what these new cells do in
Humans, a recent study in rats found the newborn neurons were crucial for
forming certain kinds of memories.
The neurogenesis hypothesis about depression is both intriguing and somewhat
unsettling. Malberg says she received an e-mail from a man who worried that
since cancer is basically an increase in cell proliferation, [drug-induced
neurogenesis] could be a bad thing, and we need to investigate this very
carefully.
Although Malberg doesn't think antidepressants will cause
cancer, she believes her findings should give people pause. I think you
have to accept that there is a structural change in your brain when you take
drugs like Prozac. If people aren't comfortable with that, that's something else
to consider.
Jacobs doesn't see cause for worry, because the new cells
seem to degenerate if they're not used.

Harvard psychiatrist Joseph Glenmullen finds such brain-altering effects more unsettling than intriguing. Last year he published Prozac Backlash: Overcoming the Dangers of Prozac, Zoloft, Paxil, and Other Antidepressants with Safe, Effective Alternatives, a book that details his brief against the drugs: They cause far more serious and common side effects than their manufacturers report; the Food and Drug Administration has failed to sufficiently investigate these reports; patients' complaints about the drugs are largely ignored; and the drugs are prescribed too often and for far too broad a range of distress. Perhaps most important, Glenmullen believes the way the drugs are marketed suggests that depression is primarily a biological problem to be solved by biochemical means, instead of a complex biopsychosocial phenomenon that can be resolved in many cases with traditional psychotherapies and without drugs. Glenmullen, who does prescribe serotonin enhancers when he deems it appropriate, likens them to such stimulants as amphetamines and cocaine—drugs that were once used widely, without fear of side effects, to give people more energy, improved mood, and increased focus.
Glenmullen has long suspected that drugs that alter serotonin metabolism cause profound changes in the brain. He bases his suspicion on a body of research during the last 20 years by scientists investigating another class of drugs that includes MDMA (Ecstasy) as well as fenfluramine, the diet drug recently removed from the market because of its association with heart valve problems. These drugs do more than just block serotonin reuptake; they primarily stimulate the release of large quantities of serotonin from nerve endings into the brain. The resulting flood is thought to cause the mind-altering effects of MDMA. And that flood, some scientists argue, leaves brain damage in its wake. When monkeys and rats are given high doses of serotonin releasers—up to 40 times the dose that people generally take—the microscopic architecture of their brains looks different from normal brains. The nerve fibers (axons) that carry serotonin to the target cells seem to change their shape and diminish in number—effects some scientists claim are properly understood as brain damage.
Glenmullen is convinced these results raise questions about other serotonergic drugs like Prozac, and a recent study has only increased his concern. Research conducted by neurologist Madhu Kalia at Jefferson Medical College in Philadelphia and scientists at the Centers for Disease Control and Prevention showed that the rats given very high doses (up to 100 times the Human dose, by body weight) of Prozac and Zoloft contained the same kinds of brain abnormalities—neurons with swollen or kinked tips—as rats who were given high doses of serotonin releasers.
Jim O'Callaghan, a Centers for Disease Control neuroscientist and a coauthor
of the study, doesn't think the results indicate that Prozac causes brain
damage. To the contrary, he and his team believe that neither serotonin
enhancers nor serotonin releasers are properly understood as neurotoxic.
According to O'Callaghan, the point of the study was to show that even a drug
like Prozac, which virtually no one claims is neurotoxic, can produce some of
the same abnormalities as the serotonin releasers. Other scientists, in his
view, have been too quick to deduce what they think is going on in the
[nerve] fibers
from two pieces of data: The serotonin releasers deplete
serotonin, and the microphotographs of brains exposed to high doses of these
drugs look abnormal. O'Callaghan believes that scientists should rethink their
definition of neurotoxicity, because high doses of Prozac and Zoloft, which do
not deplete serotonin, cause the same transient abnormalities as do high doses
of drugs such as MDMA. (Blair Austin, a spokesperson for Eli Lilly, producer of
Prozac, points out that the abnormalities have not been linked to any
physiological result. Moreover, he says, based on the high dosage and other
conditions of the study, the findings are only of minor toxicological
importance and pose no risk to Human safety.
)
The perhaps surprising fact that scientists don't agree on what constitutes
brain damage shouldn't, according to Glenmullen, distract us from what he thinks
are the crucial implications of this study. I'm not saying that Prozac is
neurotoxic,
he told me. But it should be public policy with a
neurotransmitter booster to look for neurotoxicity. And if that information is
out there, the people ought to have it.

In Glenmullen's view, regulatory agencies don't always do enough to help
consumers either. He devoted a chapter in his book to the FDA's decision to
allow Lilly not to include a warning with Prozac that the drug can cause or
worsen suicidal symptoms—despite studies that indicated that up to 3.5 percent
of patients might experience such effects. Add the advertising campaigns by the
drug companies, he says, and you have a social climate in which everyone
wants a serotonin booster
and everyone believes in a pharmacological
fantasy
that we can use mood-altering drugs for a variety of ills without
giving serious thought to the potential danger.
Glenmullen offers a different Rx: fewer drugs and more therapy. He believes many people taking serotonin-enhancing drugs would respond as well to talk therapy. And talk isn't the only option. Aerobic exercise, such as jogging or dance, also combats less severe cases of depression. Studies in rats suggest that exercise boosts serotonin and neurogenesis as well.
Of course the use of any drug, especially one that tinkers with the brain's machinery, involves risk, the full extent of which can't be known until a large number of people have used it for many years. This familiar caution may take on a new urgency when we realize that research about serotonin enhancers still offers more questions than answers. On the other hand, as we learn more about the brain's extraordinary plasticity, about the complex intertwining of neurochemistry with emotion, cognition, and experience, we may well become more comfortable with the idea of modifying our brains intentionally. We may then wish to have serotonin enhancers among the methods at our disposal to do so.